Was sind Huminsäuren?
1. Welche Vorteile haben Huminsäuren bei Umweltanwendungen?
Huminstoffe entstehen durch die chemische und biologische Humifizierung von pflanzlichen und tierischen Stoffen und durch die biologischen Aktivitäten von Mikroorganismen. Das biologische Zentrum, der Hauptanteil der natürlichen Huminstoffe, sind die Huminsäuren, die Huminsäuren und Fulvinsäure enthalten. Huminsäuren sind eine ausgezeichnete natürliche und organische Methode, um Pflanzen und Böden mit einer konzentrierten Dosis an essentiellen Nährstoffen, Vitaminen und Spurenelementen zu versorgen. Sie sind komplexe Moleküle, die auf natürliche Weise in Böden, Torf, Ozeanen und Süßwasser vorkommen. Die beste Quelle für Huminsäuren sind die Sedimentationsschichten aus Weichbraunkohle, die als Leonardit bezeichnet werden. Hier sind Huminsäuren in hoher Konzentration zu finden. Leonardit ist eine organische Substanz, die den Zustand der Kohle nicht erreicht hat und sich von Weichbraunkohle durch ihren hohen Oxidationsgrad, der durch den Prozess der Kohlebildung (Moor>Torf>Kohle) und den hohen Huminsäurengehalt sowie durch höhere Carboxylgruppen unterscheidet.
Im Vergleich zu anderen organischen Produkten ist oxidierte Braunkohle sehr reich an Huminsäuren. Während oxidierte Braunkohle das Endprodukt eines Humifizierungsprozesses von mehreren Millionen Jahren ist, ist beispielsweise die Entstehungszeit von Torf innerhalb von nur wenigen tausend Jahren abgeschlossen.
2. Vorteile von Huminsäuren bei Umweltanwendungen
Huminstoffe im Allgemeinen und Huminsäuren im Detail interagieren mit allen Klassen von Ökotoxika, einschließlich: Schwermetalle, Erdöl und chlorierte Kohlenwasserstoffe, Pestizide, nitroaromatische Sprengstoffe, Azofarbstoffe, Actinide, etc.
Huminsäuren sind dafür bekannt, stabile Komplexe mit Schwermetallen und Addukten mit hydrophoben organischen Verbindungen zu bilden; Ladungsübertragungskomplexe herzustellen; als Elektronen-Shuttles zu fungieren und Redoxreaktionen von Übergangsmetallen, von chlorierten und nitrierten Kohlenwasserstoffen zu vermitteln; an mineralischen Oberflächen zu adsorbieren; und die Interphasenverteilung der Schadstoffe zu beeinflussen. Schließlich kann die Huminstoffe die Widerstandsfähigkeit lebender Organismen gegen unspezifische Stressfaktoren stärken.
Diese einzigartige Konstellation von reaktiven Merkmalen legt nahe, dass Huminstoffe das Potenzial haben, ein breites Spektrum von Bedürfnissen innerhalb des Schwerpunktbereichs der Umweltsanierung zu erfüllen. Diese theoretische Aussage wird durch mehrere Beispiele für konkrete Anwendungen bei der Sanierung bestätigt. Um eine optimale und systematische Anwendung zu gewährleisten, ist jedoch eine erweiterte Wissensbasis über die Wechselwirkungen zwischen Humus, Ökotoxikanten und lebenden Organismen erforderlich.
Mehrere Wechselwirkungen zwischen Huminstoffen/Humfsäuren, Ökotoxika und lebenden Organismen können organisiert werden:
- bindende Wechselwirkungen, die die chemische Speziation und Bioverfügbarkeit von Ökotoxika beeinflussen;
- sorptive Wechselwirkungen, die die physikalische Speziation oder die Interphasenaufteilung von Ökotoxika beeinflussen;
- abiotisch-biotische Redox-Wechselwirkungen, die sich auf Stoffwechselwege auswirken, die mit anecotoxischen Mitteln gekoppelt sind; und,
- direkte und indirekte Wechselwirkungen mit verschiedenen physiologischen Funktionen lebender Organismen.

Abb.1: Huminstoffe können an protolytischen, Ionenaustausch- und Komplexationsreaktionen teilnehmen; an Donor-Akzeptor-Interaktionen teilnehmen; an Wasserstoffbindungen teilnehmen; und an Van-der-Waals-Interaktionen teilnehmen.
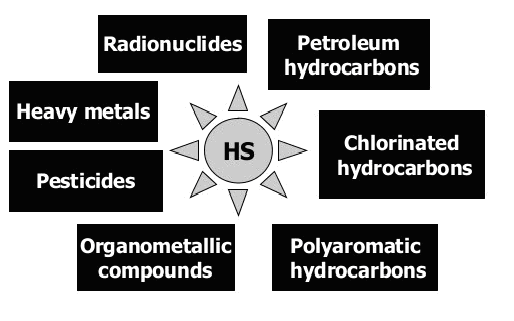
Abb. 2: Aufgrund der unterschiedlichen Reaktivität von HS können sie mit allen Klassen von Ökotoxika (ET) in der belasteten Umgebung interagieren. Huminstoffe sind dafür bekannt, stabile Komplexe mit Schwermetallen und Radionukliden zu bilden; Addukte und Ladungsübertragungskomplexe mit hydrophoben organischen Verbindungen herzustellen; Redoxreaktionen von Übergangsmetallen, chlorierten und nitrierten Kohlenwasserstoffen zu vermitteln.
(aus: I.V. PERMINOVA, K. HATFIELD, REMEDIATION CHEMISTRY OF HUMIC SUBSTANCES: THEORIE UND IMPLIKATIONEN FÜR DIE TECHNOLOGIE)

